Determination by UV spectrophotometry of acetylcholine bromide content in test-strip for measuring whole blood cholinesterase activity
-
+ English摘要:目的
建立紫外分光光度法测定全血胆碱酯酶活性测定试纸(简称测酶试纸)中溴化乙酰胆碱含量的方法,并验证该方法的可行性。
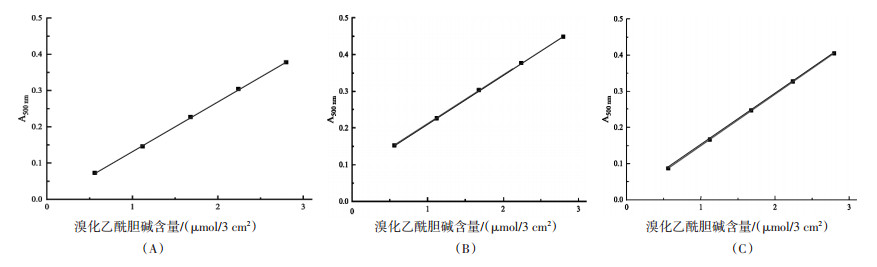
方法利用溴化乙酰胆碱和溴百里香酚蓝(测酶试纸的两种主要成分)的溶解性差异,将测酶试纸中的溴化乙酰胆碱洗脱下来,以浓度为2.8、5.6、8.4、11.2、14.0 mmol/L的溴化乙酰胆碱对照品溶液制作测酶试纸中溴化乙酰胆碱含量测定方法的标准曲线,并对该法的标准曲线线性、准确度、精密度、耐用性及回收率进行验证。
结果该方法的标准曲线线性良好,线性相关系数的平均值为0.999 79。该法的准确度、精密度和耐用性良好,批内和批间相对标准偏差均不超过4%,加样回收率在94.90%~106.22%之间。
结论该方法可用于测定测酶试纸中溴化乙酰胆碱的含量。
-
中暑是指高温环境下由于热平衡和(或)水盐代谢紊乱等引起的一种以中枢神经系统和(或)心血管系统障碍为主要表现的急性热致疾病,严重时可引起死亡 [1-2]。近30年极端高温天气频发,相关研究 [3-5]也显示上海市的中心城区、宝山区及闵行区的高温风险指数在长三角所有行政单元中位列前三,因而在当前区域开展高温中暑相关研究尤为重要。本研究拟通过收集2013—2022年每年6月15日—9月30日逐日的中暑病例信息及对应时间的相关气象信息,探讨中暑病例的流行病学特征及其与气象条件的关联强度。
1. 资料与方法
1.1 资料
以2013—2022年每年6月15日—9月30日的上海市宝山区高温中暑病例和同时期的每日气象信息作为本次研究的资料。
1.2 方法
1.2.1 中暑病例资料的收集
中暑病例资料来源于宝山区各医疗机构在中国疾病预防控制信息系统中上报的信息,病例资料主要包括性别、年龄、职业、中暑类型、中暑时间等。
1.2.2 气象资料的收集
所有气象信息来源于上海市气象局,包括宝山区历年6月15日—9月30日的每日最高气温、最低气温、平均气温、相对湿度、降水和风速等。据中国气象局规定,高温日的定义为日最高气温≥ 35 ℃,该气温连续出现3 d及以上的天气定义为高温热浪 [3]。
1.2.3 中暑预警气温参考值
在纳入本地区2013—2022年全部中暑病例后,按照不同风险计算了两种中暑风险的气温预警值,即中暑高风险气温预警值和中暑超高风险气温预警值。将中暑高风险气温预警值定义为已引发本地区5%中暑病例的气温气象条件,在此条件下有较高风险会发生中暑;将中暑超高风险气温预警值定义为已引发本地区95%中暑病例的气温气象条件,在此条件下有极高风险发生中暑。
1.2.4 质量控制
高温中暑病例由辖区内各级医疗机构临床医生诊断和报告,区疾控专员经过上级培训后对每例中暑病例进行逐一审核上报。
1.2.5 统计学分析
使用SAS 9.0软件进行统计学分析,频数分布用于人口学信息的描述、中暑类型的分型以及气象资料中高温、热浪、降雨发生与否的描述;符合正态分布的计量资料如气温、相对湿度和风速等采用均数±标准差(x ± s)表示;纳入所有中暑病例中暑当日的气温气象条件后,计算两种中暑预警气温参考值(95%置信区间),中暑高风险气温预警值=气温气象条件均值-1.64 ×气温气象条件标准差,中暑超高风险气温预警值=气温气象条件均值+ 1.64 ×气温气象条件标准差;采用等级logistic回归模型估计不同气象条件与高温中暑发生的关联强度。P < 0.05为差异有统计学意义。
2. 结果
2.1 中暑病例基本情况
本次收集的2013—2022年宝山区的中暑病例共计173例,其中男性149例(占86.1%),女性24例(占13.9%);中暑病例的平均年龄为(45.08 ± 17.85)岁,最小18岁,最大91岁,其中50岁以下的病例数共计100例(占57.8%),60岁以上的病例40例(占23.1%);中暑人群中排名前四的职业分别为建筑工人(62例,占35.8%)、离退休人员(27例,占15.6%)、物流配送人员(9例,占5.2%)和环卫工人(8例,占4.6%);有115例(占66.5%)中暑发生的地点在室外,85例(占33.5%)发生在室内。
2.2 中暑发生时间和临床表现分型
173例中暑病例中,有114例(占65.9%)发生在7月;在临床分型上,轻症中暑82例(占47.4%),重症中暑91例(占52.6%)。在重症中暑病例中热射病病例最多,共45例,占重症中暑病例总数的49.4%。见表 1。
表 1 中暑发生时间和临床表现分型项目 中暑人数 构成比/% 中暑发生时间 6月(半月) 20 11.6 7月 114 65.9 8月 37 21.4 9月 2 1.2 中暑类型 轻症中暑 82 47.4 重症中暑 91 52.6 重症中暑类型 热痉挛 19 20.9 热射病 45 49.5 热衰竭 15 16.5 混合型 12 13.2 2.3 宝山区2013—2022年监测期间气象信息
对所收集的气象数据按照平均气温、平均相对湿度、平均风速、年累计高温日、热浪日和降雨日做逐年统计,结果发现每年气象条件不尽相同,其中高温日和热浪日在2013年、2017年和2022年相对较多。见表 2。
表 2 宝山区2013—2022年监测期间气象条件年份 平均气温/℃ 高温日/d 热浪日/d 平均湿度/% 平均风速/(m/s) 降雨日/d 2013 29.2 ± 3.5 47 31 63.7 ± 10.0 1.1 ± 0.3 44 2014 26.3 ± 2.5 8 1 80.6 ± 7.5 1.0 ± 0.3 58 2015 26.6 ± 3.2 12 10 77.2 ± 11.4 0.5 ± 0.2 50 2016 28.3 ± 2.9 17 10 75.1 ± 10.0 2.3 ± 0.6 43 2017 28.8 ± 3.3 31 18 73.3 ± 10.1 2.3 ± 0.8 52 2018 28.3 ± 2.4 10 4 77.1 ± 8.1 2.8 ± 1.0 41 2019 27.0 ± 2.8 12 6 81.1 ± 8.1 2.3 ± 0.9 48 2020 26.8 ± 3.3 20 9 79.8 ± 10.8 2.3 ± 0.8 22 2021 27.6 ± 2.1 10 5 84.7 ± 9.5 2.6 ± 1.3 56 2022 28.9 ± 3.6 40 23 71.9 ± 8.6 2.6 ± 0.8 37 2.4 中暑人群与中暑气象因素的关联强度
将当日高温中暑发生情况设置为响应变量,按高温中暑病例临床严重程度划分为3组:无高温中暑病例发生、仅发生轻症中暑病例和出现重症中暑病例,分别赋值为0、1、2;将日平均气温(连续变量)、日最高气温(连续变量)、日最低气温(连续变量)、相对湿度(连续变量)、风速(连续变量)、是否降雨(二分类变量)、是否发生热浪(二分类变量)、是否为高温日(二分类变量)作为预测变量,利用多分类等级logistic回归模型分析各气象条件与高温中暑的关联强度。等级logistic回归分析结果显示:除风速外,日平均气温、日最高气温、日最低气温、相对湿度、是否降雨、是否发生热浪、是否为高温日均与中暑的发生存在着统计学关联,其中相对湿度每升高1%,出现中暑的风险降低至原来的0.901倍;相较于不发生降雨,发生降雨时出现中暑的风险降低至0.340倍;日平均气温、日最高气温和日最低气温每升高1 ℃,出现中暑的风险分别升高至原来的2.005倍、1.736倍和1.906倍;热浪和高温日时发生中暑的风险相较于非热浪和非高温日分别升高至11.896倍和15.819倍;移动3 d平均气温(即当日和前两日气温均值)中,日平均气温、日最高气温、日最低气温每升高1 ℃,出现中暑的风险分别升高至原来的1.926倍、1.699倍和1.957倍。以上差异均有统计学意义(均P < 0.001)。见表 3。
表 3 中暑与各气象因素的关联强度项目 β值 SE值 wald χ2值 P值 OR(95%CI)值 相对湿度 -0.105 0.011 84.532 < 0.001 0.901
(0.881 ~ 0.921)风速 0.070 0.094 0.553 0.457 1.073
(0.892 ~ 1.290)降雨(以不降雨为对照) -1.079 0.249 18.836 < 0.001 0.340
(0.209 ~ 0.553)热浪(以非热浪为对照) 2.476 0.237 109.241 < 0.001 11.896
(7.477 ~ 18.926)高温(以非高温为对照) 2.761 0.250 122.137 < 0.001 15.819
(9.694 ~ 25.815)当日气温 日平均气温 0.695 0.063 122.286 < 0.001 2.005
(1.772 ~ 2.268)日最高气温 0.552 0.050 121.003 < 0.001 1.736
(1.574 ~ 1.916)日最低气温 0.645 0.066 98.711 < 0.001 1.906
(1.675 ~ 2.169)移动3 d平均气温 日平均气温 0.655 0.062 112.17 < 0.001 1.926
(1.706 ~ 2.174)日最高气温 0.530 0.049 116.45 < 0.001 1.699
(1.543 ~ 1.871)日最低气温 0.671 0.072 87.002 < 0.001 1.957
(1.700 ~ 2.254)2.5 研究区域的中暑预警气温参考值
纳入全部中暑病例发生中暑当日的日平均气温、日最高气温、日最低气温3种气温气象条件后,分别计算3种气温气象条件所对应两种预警参考值即中暑高风险气温预警值和中暑超高风险气温预警值。结果显示,研究区域的中暑超高风险气温预警值的日平均气温、日最高气温、日最低气温分别为35.55 ℃、40.58 ℃和31.07 ℃,中暑高风险气温预警值的日平均气温、日最高气温、日最低气温分别为28.87 ℃、33.31和25.21 ℃。具体结果见表 4。
表 4 研究区域的中暑预警气温参考值项目 中暑超高风险气温预警值 中暑高风险气温预警值 日平均气温/℃ 35.55 28.87 日最高气温/℃ 40.58 33.31 日最低气温/℃ 31.07 25.21 3. 讨论
在本次研究所收集的中暑病例中,男性149例(占86.1%)多于女性24例(占13.9%),可能与男性更多地从事户外高强度体力劳动有关,这也与上海市金山区的研究结果 [6]一致。从年龄来看,该地区中暑病例平均年龄为(45.08 ± 17.85)岁,其中50岁以下的病例占57.8%,以青壮年劳动人口为主。该地区的中暑病例大多数发生在7月为114例(占65.9%),这与淄博、南通、宁波等地的研究 [7-10]一致,可能是由于在高温初来时机体尚未形成热适应所造成 [6]。
在2013—2022年中,该地区的平均气温、高温日天数以及热浪天数均以2013年最高,随后分别为2022年和2017年,其余年份差距不大。等级logistic回归分析结果显示,中暑的发生与日平均气温、日最高气温和日最低气温为正相关关系,每升高1 ℃,发生中暑的风险提高至1.736 ~ 2.005倍,到达高温或热浪天气时中暑发生的风险提高至11.896 ~ 15.819倍,提示当气温升高时,特别是达到高温或热浪天气时,应及时预警,提醒用人单位和劳动者做好防暑降温工作,避免高温中暑的发生。
等级logistic回归分析结果还显示,移动3 d平均气温(即当日和前两日气温均值)中,其日平均气温和日最高气温与中暑的关联强度略小于当日的日平均气温和日最高气温,但其日最低气温与中暑的关联强度大于当日,说明持续较长时间的相对较高的日最低气温更易导致中暑发生,与宁波市的研究 [11]一致。
此外,本研究还发现,中暑的发生与相对湿度为负相关关系,降雨可降低中暑发生的风险。这可能与降雨时气温相对较低,而相对湿度也相对较高有关。
当前高温预警由气象部门发布,分为黄色、橙色和红色预警3个等级,分别代表连续3 d日最高气温将在35℃以上、24 h内最高气温将升至37 ℃以上以及24 h内最高气温将升至40 ℃以上,均仅对日最高气温作出了预警。本研究通过纳入区域内所有中暑病例发生中暑当日的3种气温气象条件,制定了本地区的中暑预警气温参考值,分为中暑高风险气温预警值(纳入5%的中暑人群)和中暑超高风险气温预警值(纳入95%的中暑人群)2种。可根据实际不同情况使用灵敏度更高的高风险气温预警值,或特异度更高的中暑超高风险气温预警值,并提前发布预警,提醒公众注意防范中暑,提高人们的自我保护意识;也可根据预警值,制定针对老年人、儿童或有基础疾病人群的特别保护措施;同时也可为相关政策的制定如排定高危职业高温天的工作、制定与气温相关的劳动法规或为制定高温天气应急方案提供科学依据。
综上,该地区高温中暑的发生有以下一些特征,日平均气温、日最高气温和日最低气温的升高均会增加高温中暑发生的可能性,此效应在热浪和高温来袭时会更明显。每年7月是高温中暑的最高发时期,且男性人数远大于女性,持续3 d的相对较高的日最低气温更易导致中暑发生。高温中暑重在预防,通过了解这些特点,利用传统媒体以及新媒体提前做好高温和热浪的预警,加强对公众的健康教育,高温天气期间应注意避日晒、保睡眠、多饮水,积极预防高温中暑的发生。
本次研究不足之处主要在于仅纳入了外环境气象条件,缺少个人详细暴露情况;另外,目前响应变量仅考虑了中暑的程度,而没有考虑人数等其他方面。未来可增加收集相关基础资料以充实后续研究。
-
表 1 标准曲线系列样品批间精密度与准确度验证结果
溴化乙酰胆碱标准曲线系列含量/ (μmol/3cm2) 溴化乙酰胆碱测定值/(μmol/3 cm2) RSD/% 相对误差/%a 1 2 3 均值 标准差 0.56 0.567 0.557 0.557 0.560 3 0.005 4 0.97 0.05 1.12 1.099 1.113 1.118 1.1103 0.0100 0.90 -0.87 1.68 1.691 1.691 1.685 1.689 0 0.003 6 0.21 0.53 2.24 2.255 2.250 2.245 2.249 9 0.004 8 0.21 0.44 2.80 2.789 2.789 2.794 2.790 4 0.003 1 0.11 -0.34 [注] a相对误差=(测定值-理论值)/理论值× 100% 表 2 批内精密度与准确度验证结果
理论值/(μmol/3 cm2) 溴化乙酰胆碱测定值(/ μmol/3 cm2) RSD/% 相对误差/% 1 2 3 4 5 6 平均值 标准差 1.12 1.138 1.130 1.129 1.126 1.126 1.129 1.129 6 0.004 3 0.38 0.86 1.68 1.700 1.696 1.691 1.695 1.701 1.702 1.697 5 0.004 3 0.26 1.04 2.24 2.250 2.250 2.252 2.247 2.255 2.254 2.251 4 0.003 1 0.14 0.51 表 3 批间精密度与准确度验证结果
理论值/(μmol/3 cm2) 批次 溴化乙酰胆碱测定值(/ μmol/3 cm2) RSD/% 相对误差/% 1 2 3 4 5 6 平均值 标准差 1.12 1 1.138 1.130 1.129 1.126 1.126 1.129 1.094 8 0.040 3.66 -2.25 2 1.046 1.052 1.036 1.032 1.044 1.039 3 1.106 1.129 1.125 1.112 1.106 1.102 1.68 1 1.700 1.696 1.691 1.695 1.701 1.702 1.656 3 0.039 2.35 -1.41 2 1.608 1.602 1.603 1.602 1.610 1.614 3 1.661 1.665 1.672 1.665 1.662 1.665 2.24 1 2.250 2.250 2.252 2.247 2.255 2.254 2.228 2 0.018 0.80 -0.53 2 2.221 2.219 2.218 2.209 2.207 2.210 3 2.228 2.221 2.215 2.213 2.213 2.225 表 4 测酶试纸溴化乙酰胆碱含量检测法加样回收率检测结果
理论值/(μmol/3 cm2) 溴化乙酰胆碱回收率/% 1 2 3 4 5 均值 标准差 1.1 96.25 96.25 97.51 97.51 97.51 97.01 0.69 1.6 94.90 99.25 106.22 98.38 99.25 99.60 4.11 2.1 97.51 99.50 98.84 98.84 102.16 99.37 1.72 表 5 测酶试纸溴化乙酰胆碱含量检测法耐用性验证结果
放置时间/min 溴化乙酰胆碱测定值/(μmol/3 cm2) RSD/% 1 2 3 均值 标准差 0 1.096 1.113 1.087 1.099 0.013 1.19 10 1.098 1.115 1.094 1.102 0.011 1.02 20 1.110 1.131 1.119 1.120 0.011 0.94 40 1.083 1.110 1.104 1.099 0.014 1.27 60 1.086 1.108 1.101 1.098 0.011 1.02 表 6 简化公式计算值与线性回归方程计算值比较
溴化乙酰胆碱含量/(μmol/3 cm2) 溴化乙酰胆碱含量简化公式计算值/线性回归方程计算值(均值±标准差) RSD/% 相对误差/% 1.12 0.994 7 ±0.041 4 4.16 -0.53 1.68 0.985 2 ± 0.027 8 2.82 -1.48 2.24 0.980 5 ±0.021 6 2.21 -1.95 -
[1] 石汉文, 佟飞, 田英平.急性有机磷中毒的规范化治疗[J].中华急诊医学杂志, 2005, 14(4):351-352. doi: 10.3760/j.issn:1671-0282.2005.04.031 [2] 石敦义, 余锋, 王鹏, 等.规范性治疗急性有机磷农药中毒患者75例分析[J].临床急诊杂志, 2019, 20(1):69-71. http://www.cnki.com.cn/Article/CJFDTotal-ZZLC201901015.htm [3] 荆玉霞.血液滤过抢救急性重度有机磷中毒的临床疗效观察[J].中国全科医学, 2017, 20(2):92-93. http://www.cnki.com.cn/Article/CJFDTotal-QKYX2017S2033.htm [4] 林永春, 张玉雄, 张炎安.重症有机磷农药中毒者126例急诊救治临床分析[J].临床合理用药, 2018, 11(11A):74-75. http://d.old.wanfangdata.com.cn/Periodical/lchlyyzz201831041 [5] 冯书芳, 邱泽武.有机磷中毒救治药物研究及其临床应用[J].中国药理学与毒理学杂志, 2016, 30(12):1431-1438. doi: 10.3867/j.issn.1000-3002.2016.12.022 [6] 孙自国, 吴嘉荔, 杨立山, 等.急性有机磷农药中毒机制及治疗的研究进展[J].临床急诊杂志, 2018, 19(6):419-422. http://d.old.wanfangdata.com.cn/Periodical/zgywjjx201301081 [7] 刘世文, 李彩玉, 张元德, 等.胆碱酯酶活性的测定与临床应用[J].齐鲁医学检验, 2004, 15(5):54. http://d.old.wanfangdata.com.cn/Periodical/jkr200824014 [8] 王明山, 胡静雪, 吴兴贵, 等.急性有机磷中毒救治过程中影响胆碱酯酶活力因素[J].华北煤炭医学院学报, 2000, 2(6):624-625. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK200000858603 [9] 国家药典委员会.中华人民共和国药典(2015年版四部)[M].北京:中国医药科技出版社, 2015. -
期刊类型引用(1)
1. 庄小舟,蔡小璇,彭建梅,马争,叶立和,姚莞睿. 空气中1, 2-二氯乙烷气体检测管的应用评估. 中国工业医学杂志. 2020(06): 546-549+577 .  百度学术
百度学术
其他类型引用(0)





 下载:
下载: